Pharmaceutical Specialists
We have over 60 years of experience in handling and re-export of pharmaceuticals.
Our organization, infrastructure, processes, and systems have been specifically developed to ensure compliance with the highest standards of quality and Good Manufacturing Practices and Distribution (GMP and GDP). Our facilities are regularly audited by local authorities and by each of our clients. This makes us committed not only to local regulations, we are also committed to complying with each of the quality standards of our customers. We have a team of engineers and staff engaged on ongoing internal audit, train staff and document changes quality.
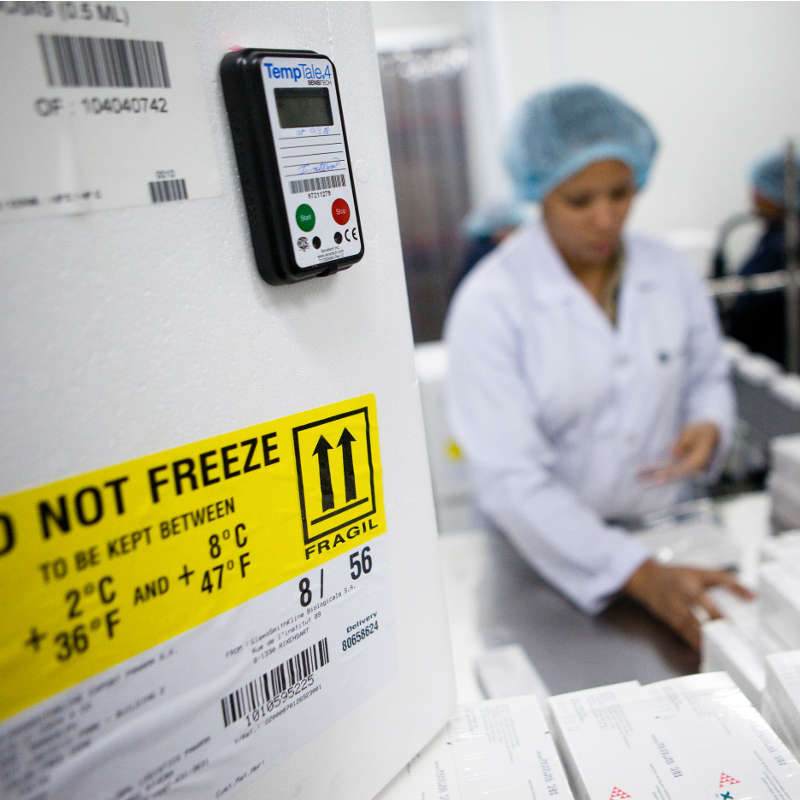
Specialist in:
- Injectable vaccines and medicines in controlled cold rooms
- Door-to-door cold chain operations
- Management of cold chain materials: boxes, gel packs, TT4’s.
- Administration validations
Specialists in Cold Chain Management:
Animal Health Specialists:
- Specific and segregated storage for animal health products.
- Cold rooms dedicated to animal health
Specialists Nutritional Products:
- Management requirements on food authority.
- Specific areas of warehouse
Infrastructure & resources:
- 22 rooms conditionings under standard “Dat and Pollution Free”
- Validated printing equipment for health records, markings, and labels
- Human Resource specialist with experience and training
- Monitoring and planning work orders for full visibility and tracking of work in progress
- Batch Record documents complete with GMP
- Quality sampling based on quality standards
- Quality inspectors on site to release jobs
Services & Features:
WHY PANAMA? DISCOVER THE PANAMA LOGISTICS HUB AND ALL OF ITS BENEFITS

